Genetic Structuring of African Lions
This essay explores the implications of the recent paper by Moore et al. (2016) in the Journal of Heredity, describing the remarkably strong phylogeographical structuring in populations of African lions, Panthera leo. The distinctive phylogeographic records of lion populations in the Okavango and Etosha basins have an interesting bearing on the Pleistocene evolution of Africa’s palaeoenvironments. This commentary complements the OUP Tumblr Blog, and I include key references that space restrictions precluded in the original article [notably on southern African giraffes]. Please read on….And also see my recent MegaKalahari post exploring lions and hominins in Late Cenozoic wetlands….
Male lion, Serengeti-Masai ecosystem. Photographs – Mary Coulson LRPS
Much of South-central Africa is characterized by a savannah environment, with summer rainfall and dry winters. However, a number of major seasonal wetlands, such as the Okavango Delta in northern Botswana and the Bangweulu Basin in north-west Zambia, occur as “islands” within this savannah habitat. The former Palaeo-Lake Etosha was also a major representative of this wetland archipelago until it lost the sustaining inflow of the Kunene river. This wetland mosaic has persisted in feature of south-central Africa for at least the past 5 million years, although their locations and extent have changed over this time period. Major rearrangements in the links among rivers across the south-central Africa have also controlled the extent of these inland drainage sumps. In the same evolutionary timeframe, these dynamic river links also enabled and contained dispersals of aquatic and savannah species, respectively.

Okavango lionesses resting after feasting on a zebra kill. The young lion with them is cooling off in a pool of water. Photograph – Andy Moore
There is interesting evidence that the vast wetlands of the African hinterland have provided important refugia for biodiversity, notably mammal and bird species, and particularly over recurring episodes of extreme aridity through the past 3 million years that corresponded to global ice ages. In tandem with the dynamics of rivers feeding and thus linking these drainage basins, climatic oscillations likely explain why these wetland “islands” in the vast savannahs host a diversity of wetland specialists, notably in the Lechwe and Sitatunga antelopes. The deep evolutionary history of this wetland biota of south-central Africa is exemplified by the diverse guild of swamp-dwelling birds, including the Swamp Boubou (see phylogeny of the Laniarius shrikes in Voelker et al. 2010).
Recent genetic studies of lions in east and south-central Africa recovered significant geographical structuring of different populations across the continent [Antunes et al. 2008; Barnett et al. 2014]. These events are too old to have been caused by recent declines of the species. A most unexpected discovery is that lions in wetland habitats are genetically distinct from those of the surrounding savannah populations [Moore et al. 2016]. This implies that there has been a long-term absence of interbreeding between lions occupying these contrasting habitats. We are still unsure of how or when the wetland and savannah lion populations became genetically isolated, but hypothesize that Africa’s ancestral lion populations were wetland specialists. In contrast to these persistent wetland populations, lions isolated during Africa’s arid periods were forced to adapt to survive the expansion of deserts and arid savannahs. The outcome of new behavioural and ecological adaptations in desert and savannah lions acted to genetically isolate the derived savannah and wetland populations. This hypothesis argues further that expansion of grasslands and herbivore populations during ameliorating, wetter climatic periods. This more recent climatic event stimulated the rapid population expansion and diversification of highly successful savannah lion specialists which is evident in the genetic structuring of savannah lions in southern and east Africa. The wetland lions persist as testimony to the hidden history of the species’ evolution.
The phylogeography of Africa’s surviving giraffe populations holds interesting parallels to the complexities revealed in populations of Panthera leo. These six (at least) giraffe species also reveal significant genetic structuring across the continent, with an anomalous distributional break between the Angolan and Southern Giraffes around the Okavango delta. Equally, the phylogeography of southern African baboons, the Papio ursinus species complex, reveals strong the controls over where baboon populations have been able to persist in MegaKalahari landscapes [this Post and Paper]. This intriguing anomaly we see in northern Botswana in the evolutionary legacy of these large mammals suggests controls of the shallow rift valley of drainage evolution have also acted to isolate the two giraffe populations (Bock et al. 2014).
It is significant to the genetic structuring of wetland biodiversity that the Etosha and Okavango basins are enshrouded in the vast Mega-Kalahari sandsea (Haddon & McCarthy 2005). Today, the extent of the regional forests, woodlands, savannas and deserts of the region form a palaeoecological snapshot of a turbulent zone; since at least the Pliocene, shifts in southern hemisphere climate were magnified across the Kalahari: and especially since the Miocene. Minor increases in aridity over the Kalahari sands have magnified ecological impacts, because its austere edaphic conditions (the sandy soils and calcrete pans) dictate that only a regime of reliable, mesic rainfall can sustain woodland and forest (White 1983; Barham 2000).
Indeed, we see this in the major portion of the northern and eastern Kalahari supporting the broad forest and moist savanna belts, which cover much of western Zambia, northern Angola and the southern Congo. Rainfall is not only relatively high, but more reliable in persisting from November into May. The contrast to the deserts centered on the Kalakgadi desert in the southwest Kalahari could not be starker. It can be argued that Kalakgadi conditions dominated the subcontinent during the climatic shifts driven by the coldest, and thus most arid, Pleistocene glacials. During these arid periods, a Mega-Kalahari desert would have covered much of the subcontinent: extending from the southern Congo basin to the Orange River, and from Zambia’s Plateau in central Africa to the Angolan Escarpment against the South Atlantic.
This means the biodiversity of subcontinental Africa has endured the recurring impacts of several pervasive vegetation shifts. These have been repeated over at least the past 3 million years. This hypothesis of Mega-Kalahari Expansions and Contractions, since the Pliocene, gives us the most plausible explanation for how the wetland lions became geographically isolated as the flagship species in the wetland assemblages of the Etosha Basin and the Okavango Delta.
Two additional lines of evidence are no less significant, as they have a profound bearing on the origins of hominins in Late Cenozoic Africa – My More Recent Post.
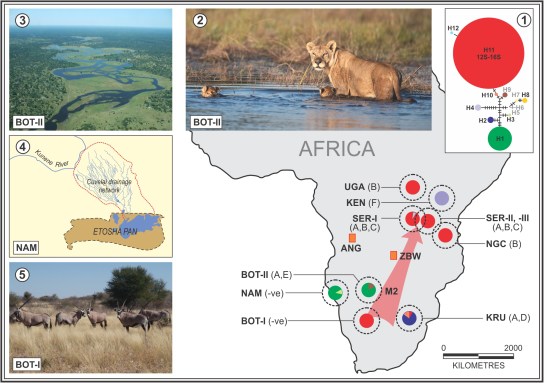
Figure 1 in Moore et al. 2016 (© The American Genetic Association 2015) – mtDNA haplotypes frequencies in African lion populations, synthesized from data presented in Antunes et al. (2008). Their phylogenetic network (inset 1) is provided primarily to identify the haplotypes documented by these authors. This should be reevaluated using more robust statistical tests, coupled with additional habitat-based genetic data, including populations from further wetland environments. Note that the isolated populations SER-II and SER-III, both characterized by a single mtDNA haplotype (H11), cannot be separated at the scale of the figure. Letters denote the FIVPle subtypes associated with each population. The pink arrow labeled M2 denotes the inferred dispersal path from the southern Kalahari (BOT-I) via the Okavango (BOT-II) into East Africa in response to expanding savannah habitats following a Pleistocene arid glacial episode. Inset 2: Lioness with swimming cubs from the Okavango (BOT-II) seasonal wetland (image courtesy of Matthew Copham of Safari Footprints; Maun, Botswana). Inset 3: Annual flood in the Okavango seasonal wetland (image courtesy of Glynis Humphrey). Inset 4: The intricate network of channels, locally known as oshanas, forming the Cuvelai seasonal wetland, which drains into Etosha Pan. Inset 5: Gemsbok (Oryx) adapted to survive in the arid savannah habitat of the southwest Kalahari (BOT-I), which lacks surface water during the winter months.
References
Antunes A, Troyer AL, Roelke ME, Pecon-Slattery J, Packer C et al. (2008). The evolutionary dynamics of the lion Panthera leo revealed by host and viral population genomics. Plos Genet. 4:1-11.
Barnett R, Yamaguchi N, Shapiro B, Ho SYW, Barnes I, Sabin R, Werdelin L, Cuisin J, Larsen G. (2014) Revealing the maternal demographic history of Panthera leo using ancient DNA and a spatially explicit genealogical analysis. BMC Evolutionary Biology 14:70.
Bock, F., Fennessy, J., Bidon, T., Tutchings, A., Marais, A., Deacon, F., & Janke, A. (2014). Mitochondrial sequences reveal a clear separation between Angolan and South African giraffe along a cryptic rift valley. BMC Evolutionary Biology, 14(1), 219.
Moore A.E., F.P.D. Cotterill, C.W. Winterbach, H.E.K. Winterbach, A. Antunes & S.J. O’Brien. (2015) Genetic evidence for contrasting wetland and savannah habitat specializations in different populations of lions (Panthera leo). Journal of Heredity 22 DOI: 10.1093/jhered/esv097
Voelker, G., Outlaw, R.K., Reddy, S., Tobler, M., Bates, J.M., Hackett, S.J., Kahindo, C., Marks, B.D., Kerbis Peterhans, J.C. and Gnoske, T.P., 2010. A new species of boubou (Malaconotidae: Laniarius) from the Albertine Rift. The Auk, 127(3), pp.678-689.




Recent Comments